A key challenge in microgravity was studying real-time gene expression. To overcome the limitations of bulky equipment like PCR, we developed a bioelectronic biochip integrating ultrasound lysis. This platform showed immediate translational potential for terrestrial diagnostics. By 2014, it had already been validated on multiple fronts: detection of HPV-16 via E6/E7 proteins, the BCR-ABL1 marker for leukemia, SARS-CoV-2 with agreement to RT-PCR, and cardiovascular biomarkers using nanoparticles. Thus, this technology not only advances space research but also establishes a new generation of portable and automated diagnostics.
BIOCHIP:
1. Human Papillomavirus (HPV):
Immobilization of molecular structures E6 and E7 on a carbon conductive substrate coated with a gold layer for the detection of HPV type 16 at the BIOTECHCELL SAC laboratory in 2024.
2. Chronic Myeloid Leukemia (CML):
Immobilization of the BCR-ABL1 gene on a carbon conductive substrate coated with a gold layer for the detection of Chronic Myeloid Leukemia at the BIOTECHCELL SAC laboratory, 2024.
3. Cardiovascular Diseases (DC):
Implementation of a diagnostic device through the immobilization of nanoparticles on a screen-printed gold electrode for the detection of cardiovascular diseases at the BIOTECHCELL SAC Laboratory in 2024
4. Diagnostic Tests for COVID-19 (SARS-CoV-2):
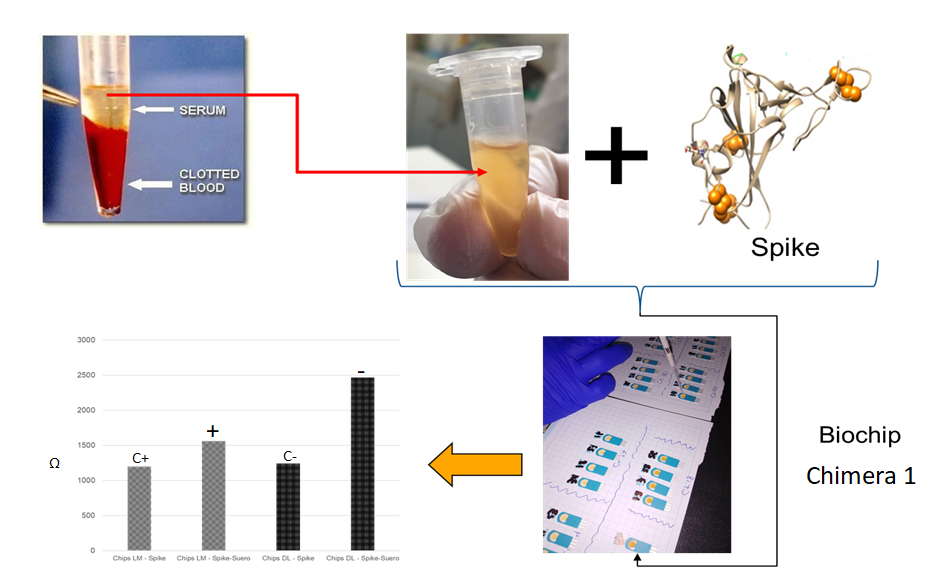
Method validation for the detection of SARS-CoV-2 using Protobiochip, compared to the RT-PCR method, in samples from COVID-19 patients.
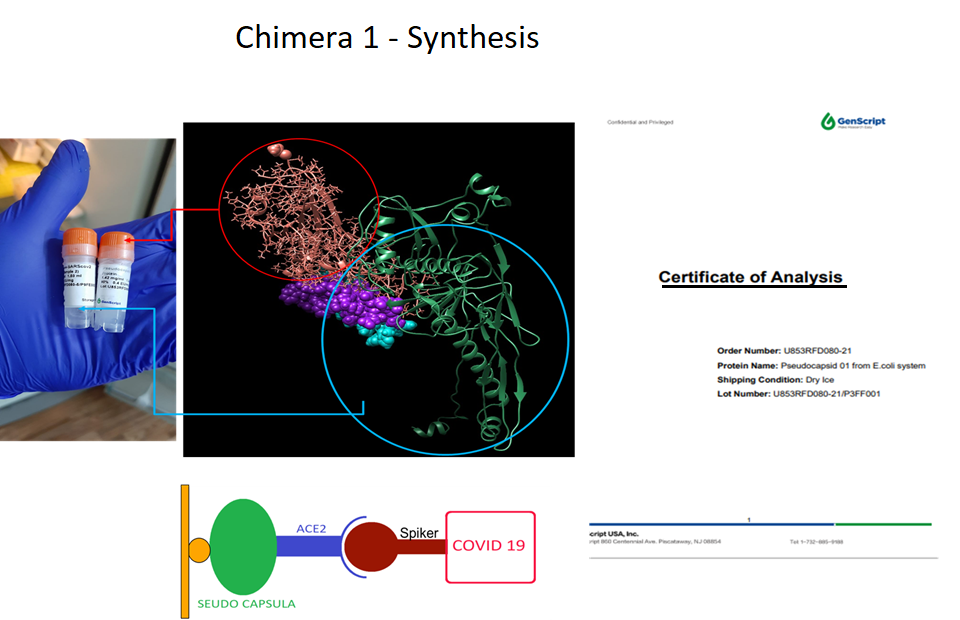
5. Diagnostic Tests for COVID-19 (SARS-CoV-2):
Method validation for the detection of neutralizing antibodies for SARS-CoV-2 using a Protobiochip, compared to the quantitative FIA test kit. Research project registered with the National Institute of Health (INS).